Biotech & Biomed Execution for R&D, GMP, and Clinical Translation
We help founders, boards and hospital groups turn science into regulated products and programs: lab build-outs, QMS/GMP, clinical development, and BD & partnerships across APAC & Europe.
Current: CSO (Miskawaan Health Group)
Former: Senior Director (Celligenics) and COO (Austrianova / SG Austria)
What We Do
Focused, senior execution you can deploy immediately.
R&D & Clinical Translation
Program strategy, protocol pathways, Phase I/II design support, KOL/CRO alignment, and clinical operations readiness.
GMP/QMS & Tech Transfer
Build or upgrade QMS/QA/QC, supplier qualification, pilot-to-GMP scale-up, documentation suites, and validation.
Lab Build-Outs & Operations
Value proposition, equipment specs, layout/flows, method validation, hiring, and operations playbooks.
Advanced Biologics & Regenerative
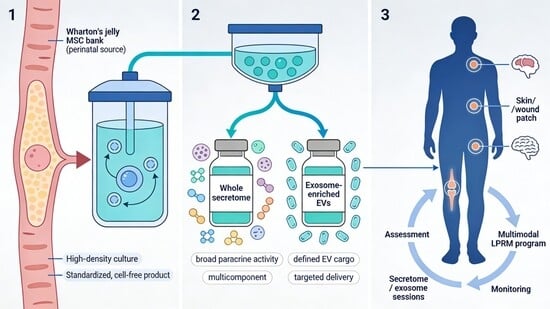
Cell therapy, secretome, exosomes, and probiotics; manufacturing partners and regulatory road-mapping.
Business Development & Partnerships
Corporate BD, licensing & co-development, investor materials, diligence, and deal support.
Interim Leadership
Hands-on COO/CSO-level execution to de-risk timelines, budgets, and stakeholder alignment.
Selected Engagements & Outcomes
- Clinical trials management — Enabled commercialization steps, QMS support, co-authored publications.
- Regenerative medicine program — Enabled commercialization steps, QMS support, co-authored publications.
- Clinical network CSO — Executive Committee, gateway for advanced therapeutics, Head of Procurement, clinical rollout.
- Cell therapy manufacturing — GMP process/device work; two oncology trials completed.
Why Anovasia
25+ years from bench to bedside; 30+ publications, 7 core inventions; APAC-EU network. Blend of science leadership + operations + BD—focused on outcomes, not slideware.
Principal

Hands-on
John A. Dangerfield, PhD — Current: CSO (Miskawaan Health Group)
Former: Senior Director (Celligenics) and COO (Austrianova / SG Austria). Specialist in cell & gene therapy, regenerative medicine, clinical development, and GMP/QMS.
Capabilities snapshot
- GMP/QMS set-up & audits
- Clinical ops & CRO/KOL alignment
- Tech transfer & scale-up
- Commercial & BD support
- Fluent in German language
- Investment & grants
Recent Publications & Media
Recent peer-reviewed work and public commentary.
Development and Functional Testing of a Novel In Vitro Delayed Scratch Closure Assay
First description of a chronic wound in vitro model using pro-inflammatory stimulation, validated with Wharton's jelly MSC secretome. Aw, Chen, Yeo, Dangerfield & Mok. Histochemistry and Cell Biology, Vol. 162.
Extracellular Vesicles and Their Membranes: Exosomes vs. Virus-Related Particles
Comparative analysis of exosome and virus-related particle biology and membrane properties. Cortes-Galvez, Dangerfield & Metzner. Membranes 13(4):397. PMID: 37103824.
Contact
Email: john@anovasia.com | LinkedIn: /john-a-dangerfield
Prefer a quick form? Drop a note below—I'll reply within one business day.